
- Chemistry - Home
- Chemistry - Introduction
- Chemistry - Branches
- Chemistry - Radioactivity
- Chemistry - Nuclear Energy
- Chemistry - Metals
- Chemistry - Metallurgy
- Chemistry - Sodium
- Chemistry - Calcium
- Chemistry - Aluminum
- Chemistry - Magnesium
- Chemistry - Maganese
- Chemistry - Iron
- Chemistry - Copper
- Chemistry - Silver
- Chemistry - Gold
- Chemistry - Platinum
- Chemistry - Zinc
- Chemistry - Mercury
- Chemistry - Plutonium
- Chemistry - Uranium
- Chemistry - Lead
- Chemistry - Thorium
- Chemistry - Hydrogen
- Chemistry - Helium
- Chemistry - Oxygen
- Chemistry - Carbon
- Chemistry - Nitrogen
- Chemistry - Chemical Law
- Chemistry - Discovery of Elements
- Elements With Their Valence
- Elements With Their Atomic Number
- Chemistry - Nobel Prize
- Chemistry Part 2 - Online Quiz
- Chemistry Part 2 - Online Test
- Chemistry Part 2 - Quick Guide
- Chemistry - Useful Resources
- Chemistry - Discussion
Chemistry - Calcium
Introduction
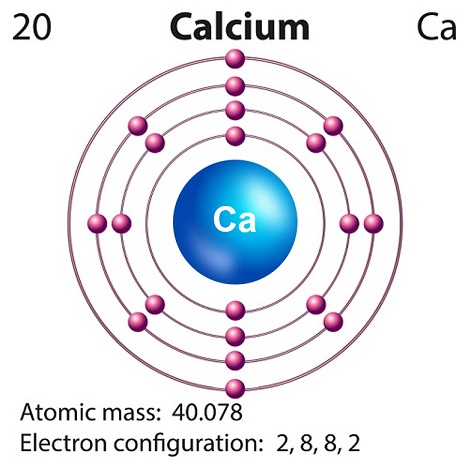
Calcium is a soft silvery-white alkaline element found largely in the Earths crust.
Calciums symbol is Ca and atomic number is 20.
Free calcium hardly exists in nature, as it is highly reactive.
Calcium is produced usually in supernova nucleosynthesis.
Salient Features of Calcium
Calcium is one of the most abundant metals by mass found in many animals.
Calcium is a very important constituent of teeth, bone, and shells.
Calcium carbonate and calcium citrate are the major dietary supplements required for a good health.
World Health Organization (WHO) listed calcium as the Essential Medicines.
In comparison to other metals, the calcium ion and most of the other calcium compounds have low toxicity.
If calcium comes in contact with water or acid, it reacts with them and becomes hazardous.
If calcium comes in contact with air, it reacts swiftly and forms a gray-white coating of calcium nitride and calcium oxide.
Most of the calcium salts are normally colorless.
When calcium burnt, the color of its flame appears brick red (see the image given below).

Calcium metal has comparatively a higher electrical resistivity than aluminum or copper.
Occurrence
Calcium occurs usually in sedimentary rocks.
The minerals (sedimentary) in which calcium found are calcite, dolomite, and gypsum.
Calcium also found in igneous and metamorphic rocks mostly in the silicate minerals, such as amphiboles, plagioclases, pyroxenes, and garnets.
Calcium also found in many of the food products namely dairy products, almonds, hazelnuts, soy beans, broccoli, dandelion leaves, figs, and in many more.
Compounds of Calcium
Calcium oxide - CaO
Calcium hydroxide - Ca(OH)2
Calcium chloride - CaCl2
Calcium hypochlorite (Bleaching powder) - Ca(ClO)2
Calcium phosphate - Ca3(PO4)2
Uses of Calcium
Calcium has wide range of usage, significant of them are −
Calcium carbonate (CaCO3) is used in manufacturing cement.
Calcium carbonate (CaCO3) is also used in making toothpaste.
In insecticides, calcium arsenate (Ca3(AsO4)2) is used.
Calcium chloride (CaCl2) is used in ice removal as well as in dust control.
Calcium citrate (Ca3(C6H5O7)2) is commonly used as food preservative.
Calcium gluconate (Ca(C6H11O7)2) is used frequently as a food additive as well as in vitamin pills.
Calcium hypochlorite (Ca(OCl)2) is generally used as a swimming pool disinfectant, as a bleaching agent.
