
- Chemistry - Home
- Matter In Our Surroundings
- Is Matter Around Us Pure
- Chemistry - Atoms & Molecules
- Chemistry - Structure Of The Atom
- Chemical Reactions and Equations
- Chemistry - Acids, Bases, and Salts
- Materials: Metals and Non-Metals I
- Chemistry - Metals & Non-Metals II
- Carbon & its Compounds
- Periodic Classification Of Elements
- Synthetic Fibres and Plastics
- Chemistry - Coal And Petroleum
- Chemistry - Combustion And Flame
- Chemistry Part 1 - Online Quiz
- Chemistry Part 1 - Online Test
- Chemistry Part 1 - Quick Guide
- Chemistry - Useful Resources
- Chemistry - Discussion
Chemistry - Matter In Our Surroundings
Introduction
Everything found in this universe is made up of some materials, scientists have named them as matter. For example, the food we eat, the air we breathe, stones, clouds, stars, plants, animals, water, dust, everything is categorized as matter.
Characteristics of Particles of Matter
Particles of matter are very small, normally, not visible from naked eye.
Particles of matter keep moving continuously, which is known as the kinetic energy.
Kinetic energy of particles directly depends on the temperature, as temperature increases, the speed of the movement also increases.
The particles of matter have attracting force; therefore, they attract each other.
The attracting force of the particles keeps the particles together; however, the strength of the attracting force varies from one kind of matter to another.
States of Matter
Matter has three following states −
Solid State
Liquid State
Gaseous State
Lets discuss them in brief −
Solid State
All the solid materials have a definite shape, distinct boundaries, and fixed volumes.
Most of the solid materials have negligible compressibility.
All the solid materials have a natural tendency to maintain their shape when subjected to outside force.
The solid materials can be broken under applied force, but it is very difficult to change their shape, as they are rigid.
Liquid State
Unlike solids, liquids have no fixed shape; however, they have a fixed volume.
Liquids take up the shape of the container in which they are kept.
Liquids have the property to flow and change shape.
Gaseous State
Matter in the form of air, which is neither solid nor liquid, is known as gas. For example, oxygen, nitrogen, hydrogen, etc.
Unlike solid, gas has not definite size and shape.
The gases, such as liquefied petroleum gas (LPG used in cooking); compressed natural gas (CNG used as fuel in vehicles), etc. have high compressibility; therefore, large volume of a gas can be compressed into a small cylinder and can be transported easily.
Gases, normally, show the property of diffusing very fast into other gases. This is the reason that we can smell (either good or bad) from the distance.
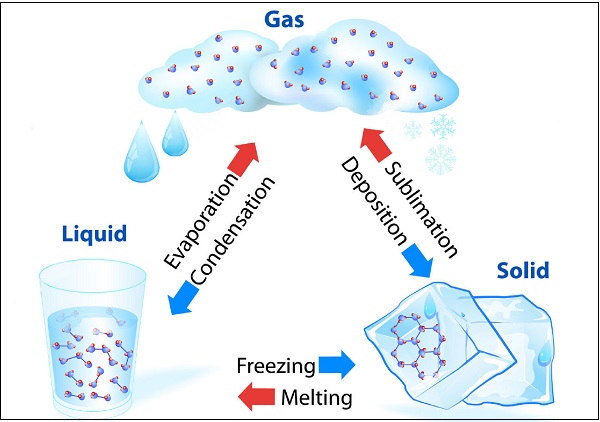
Matter Can Change its State
Water can exist in all three states, e.g. Ice as solid; water (H2O) as liquid; and water vapor as gas. The following diagram illustrates the transformation of water in different states −
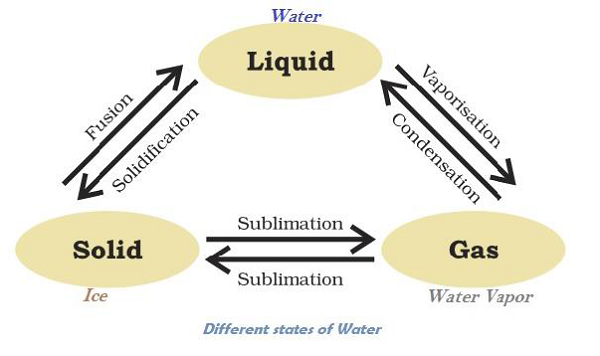
The temperature, at which solid melt and transform into the liquid (at the given atmospheric pressure), is known as melting point.
The melting point of a solid is an indication of the strength of the force of attraction between its particles.
The melting point of ice is 273.16 K, i.e. 00 C.
The process of melting (i.e. change of solid state into liquid state) is known as fusion.
The amount of heat energy, which is required to change 1 kg of a solid materials into liquid materials at a given atmospheric pressure, is known as the latent heat of fusion.
The temperature at which a liquid starts boiling at the given atmospheric pressure is known as boiling point.
The boiling point of water is 373 K i.e. 1000C.
A change of state of a matter directly from solid to gas without changing into liquid state (or vice versa) is known as sublimation.
The phenomenon i.e. change of a liquid into vapors at any temperature below its boiling point is known as evaporation.
Solid carbon dioxide (CO2) is stored under high pressure.

Solid CO2 gets converted directly into gaseous state once the pressure decreases to 1 atmosphere.
Atmosphere (atm) is a unit of measuring pressure exerted by the gas and the unit of pressure is Pascal (Pa); 1 atmosphere = 1.01 105 Pa.
The Fourth State of Matter
Plasma is the state that consists of super energetic and super excited particles.

The super excited particles are found in the form of ionized gases. E.g. the fluorescent tube (which contains helium gas) and neon sign bulbs (which contain neon gas) consist of plasma.
