Element X forms a chloride with the formula XCl2, which is solid with a high melting point. X would most likely to be in the same group of the periodic table as- Na
- Mg
- Al
- Si
Correct answer: (b) Mg
Explanation: X would most likely be in the same group of the periodic table as Magnesium (Mg).
[Extra information:
Periodic table: A periodic table is a tabular display of all the chemical elements that are arranged by the order of their atomic number, electronic configuration, and recurring chemical properties.
It displays the names, atomic numbers, symbols and atomic weights of known elements. It serves as a great tool for solving chemistry problems.
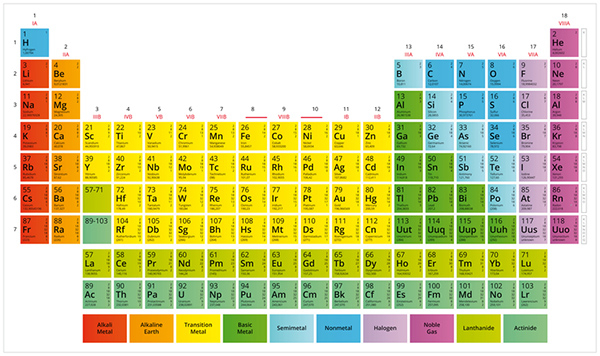
A periodic table is divided into groups (columns), where elements with each group behave similarly while bonding with other elements; and periods (rows), where elements in one period have the same number of electron shells.
The periodic table is important because it is organized to provide a great deal of information about elements and how they relate to one another in one reference. The table can be used to predict the properties of all the elements, even those that have not yet been discovered.]
Related Articles
- Element X forms a chloride with formula, XCl2, which is a solid with a high melting point. X would most likely to be in the same group of the periodic table asNaMgAlSi
- Element X' forms a chloride with the formula XCI2, which is a solid with high melting point. X would most likely be in the same group of the periodic table as:SiMgAlNa
- An element X is in group 2 of the periodic table:(a) What will be the formula of its chloride? (b) What will be the formula of its oxide?
- An element 'X' reacts with O2 to give a compound with a high melting point. This compound is also soluble in water. The element 'X' is likely to be:(a) iron (b) calcium (c) carbon (d) silicon
- A metal X is in the first group of the periodic table. What will be the formula of its oxide?
- An element X combines with oxygen to form an oxide XO. This oxide is electrically conducting.(a) How many electrons would be there in the outermost shell of the element X?(b) To which group of the periodic tables does the element X belong?(c) Write the formula of the compound formed when X reacts with chlorine
- An element X from group 2 of the Periodic Table reacts with Y from group 17 to form a compound. Give the formula of the compound.XY2XYX2Y(XY)2
- An element X from group 2 reacts with element Y from group 16 of the periodic table.(a) What is the formula of the compound formed?(b) What is the nature of bond in the compound formed?
- An element reacts with oxygen to give a compound with a high melting point. This compound is also soluble in water. The element is likely to be(a) calcium(b) carbon(c) silicon(d)iron.
- An element X forms an oxide X2O3. In which group of Mendeleev's periodic table is this element placed?(a) group II (b) group III (c) group V (d) group VIII
- A non-metal X which is the largest constituent of air combines with hydrogen when heated in the presence of iron as catalyst to form a gas Y. When gas Y is treated with sulphuric acid, it forms a compound Z which is used as a chemical fertilizer.(a) What are X, Y, and Z? (b) To which group of periodic table does X belong? (c) Name the period of periodic table in which X is placed. (d) Which element is placed just before X in the period? (e) Which element is placed just after X in the period?
- If an element X is placed in group 14, what will be the formula and the nature of bonding of its chloride?
- Reaction between X and Y, forms compound Z. X loses electron and Y gains electron. Which of the following properties is not shown by Z?(a) Has a high melting point(b) Has a low melting point(c) Conducts electricity in molten state(d) Occurs as solid
- An element ‘X’ belong to 3rd period and group 13 of the Modern Periodic Table.(a) Determine the valence electrons and the valency of ‘X’.(b) Molecular formula of the compound formed when ‘X’ reacts with an element ‘Y’ (atomic number = 8).(c) Write the name and formula of the compound formed when ‘X’ combines with chlorine.
- Carbon, Group (14) element in the Periodic Table, is known to form compounds with many elements. Write an example of a compound formed with(a) chlorine (Group 17 of Periodic Table)(b) oxygen (Group 16 of Periodic Table)
Kickstart Your Career
Get certified by completing the course
Get Started


 Data Structure
Data Structure Networking
Networking RDBMS
RDBMS Operating System
Operating System Java
Java MS Excel
MS Excel iOS
iOS HTML
HTML CSS
CSS Android
Android Python
Python C Programming
C Programming C++
C++ C#
C# MongoDB
MongoDB MySQL
MySQL Javascript
Javascript PHP
PHPPhysics
Chemistry
Biology
Mathematics
English
Economics
Psychology
Social Studies
Fashion Studies
Legal Studies


