Article Categories
- All Categories
-
 Data Structure
Data Structure
-
 Networking
Networking
-
 RDBMS
RDBMS
-
 Operating System
Operating System
-
 Java
Java
-
 MS Excel
MS Excel
-
 iOS
iOS
-
 HTML
HTML
-
 CSS
CSS
-
 Android
Android
-
 Python
Python
-
 C Programming
C Programming
-
 C++
C++
-
 C#
C#
-
 MongoDB
MongoDB
-
 MySQL
MySQL
-
 Javascript
Javascript
-
 PHP
PHP
-
 Economics & Finance
Economics & Finance
Difference between Conductor, Semiconductor, and Insulator
In order to differentiate the Conductor, Semiconductor, and Insulator, first we have to understand their extent of forbidden band i.e. separation between their conduction and valance band. The main difference between the conductor, semiconductor and insulator is in their conductivity.
Conductor
A conductor is a type of material that allows the electric current to flow through it i.e. it possesses least resistance in the path of free electrons. In case of conductor, the valance and conduction bands overlap. Due to this overlapping, a small potential difference across a conductor causes the free electrons to constitute electric current.
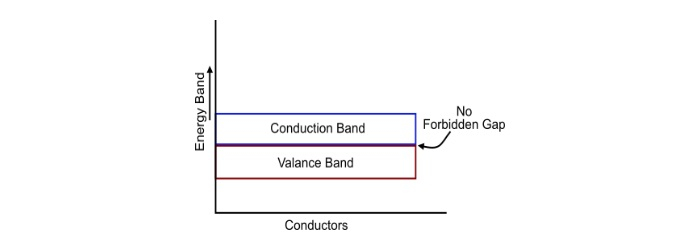
All the metals are conductors. The resistance of the conductors increases with the increase in the temperature. Hence, the conductor have positive temperature co-efficient of resistance.
The conductors are used for making conductor wires and cables for carrying electric current.
Insulator
An insulator is type of material that does not allow the electric current to pass through it, due to its high electrical resistance. In the insulators, the energy gap between valance and conduction bands is very large (about 15 eV). Therefore, a very high electric field is required to push the valance electrons to the conduction band. Due to this, there are no free electrons in the conduction band. For this reason, the electrical conductivity of insulators is very low and may considered nil under ordinary conditions.
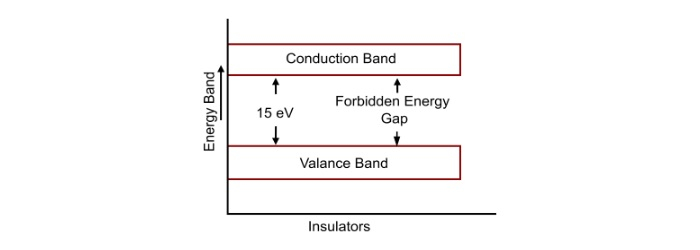
At room temperature, the valance electrons of the insulator do not have enough energy to cross over to the forbidden energy gap. But, if the temperature is raised, some of the electrons may acquire enough energy to cross over to the forbidden energy gap. Hence, the resistance of the insulator decreases with the rise in temperature. Therefore, the insulators have negative temperature co-efficient of resistance.
Due to high electrical resistance, the insulators are used for protection against electric shocks.
Semiconductor
The semiconductors are the materials having conductivity in-between conductors and insulators. In a semiconductor, the forbidden energy gap between valance and conduction bands is very small (about 1 eV) as compared to insulators. Therefore, a smaller electric field (smaller than insulators but greater than conductors) is required to push the free electrons from valance band to the conduction band.
![]()
At low temperature, the valance band of semiconductor is completely full and the conduction band is completely empty. Thus, a semiconductor behaves as an insulator at low temperature. However, at room temperature, some electrons can cross the forbidden energy gap, imparting a little conductivity to the semiconductor. As temperature is increased, more valance electrons cross over to the energy gap to reach to the conduction band and the conductivity increases. This shows that electrical conductivity of semiconductor increases with the rise in temperature. Hence, a semiconductor has negative temperature coefficient of resistance.
The conductivity of semiconductors can also be increased by adding some impurity in the pure semiconductor material, called doping. The semiconductors are commonly used in manufacturing of solid state electronic devices.
Difference between Conductor, Semiconductor and Insulator
| Parameter | Conductor | Semiconductor | Insulator |
|---|---|---|---|
| Definition | A material that allows electric current to pass through it very easily. |
A material that has conductivity in between conductors and insulators. |
Materials that do not allow the electric current to pass through them. |
| Forbidden Energy Gap | No energy gap i.e. the conduction band overlap the valance band. |
Small energy gap (approx. 1 eV). | Very large energy gap (approx. 15 eV). |
| Conductivity | High Conductivity (of the order of 10-7 mho/m). | Intermediate conductivity (ranging from 10-7 mho/m to 10-13 mho/m). | Very low conductivity(of the order of 10-13 mho/m). |
| Conduction | Due to free electrons. | Due to movement of both electrons and holes (positive charge carriers). | No conduction. |
| Resistivity | Low (of the order of 10-5Ω/m). | Intermediate (from 10-5Ω/m to 105 Ω/m). | Very high (of the order of 105 Ω/m). |
| Temperature Coefficient of Resistivity | Positive | Negative | Negative |
| Valance Electrons in Outermost Shell | Less than 4 | 4 | More than 4 |
| Examples | Metals like silver (best conductor), gold, copper, aluminium etc. | Silicon, Germanium, Gallium, Arsenide etc. | Air, Mica, Glass, Paper, Porcelain, Wood etc. |
| Application | In the manufacturing of conducting wires and cables. | In the manufacturing of solid state electronic devices like ICs, diodes, transistors etc. | Used for providing insulation electrical and electronic devices, for preventing electric shock etc. |


