Article Categories
- All Categories
-
 Data Structure
Data Structure
-
 Networking
Networking
-
 RDBMS
RDBMS
-
 Operating System
Operating System
-
 Java
Java
-
 MS Excel
MS Excel
-
 iOS
iOS
-
 HTML
HTML
-
 CSS
CSS
-
 Android
Android
-
 Python
Python
-
 C Programming
C Programming
-
 C++
C++
-
 C#
C#
-
 MongoDB
MongoDB
-
 MySQL
MySQL
-
 Javascript
Javascript
-
 PHP
PHP
-
 Economics & Finance
Economics & Finance
Liquid - Liquid Extraction
Introduction
Liquid-liquid extraction (LLE), also known as solvent extraction or partitioning, is a widely used separation technique in the chemical, pharmaceutical, and environmental industries. The technique involves the separation of a mixture of two or more liquids, based on their relative solubilities in two immiscible liquids, typically an organic solvent and an aqueous solution.
LLE can be used for a variety of purposes, including the removal of impurities, the concentration of a desired product, or the isolation of specific compounds from a complex mixture.
The given topic provides an overview of the principles, techniques, and applications of liquid-liquid extraction.
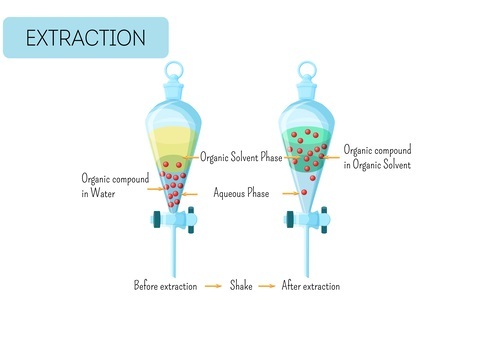
Principles of Liquid-Liquid Extraction
The principle of liquid-liquid extraction is based on the different solubilities of the components of a mixture in two immiscible liquids. The two liquids used in LLE are typically a polar solvent, such as water, and a nonpolar solvent, such as an organic solvent.
When the mixture is added to the two-phase system, the components with higher solubility in the polar solvent will remain in the aqueous phase, while those with higher solubility in the nonpolar solvent will transfer to the organic phase.
The extent of the partitioning of a component between the two phases depends on several factors, including the solubility of the component in each phase, the relative amounts of the two solvents, and the nature of the solvents themselves.
The partition coefficient, which is defined as the ratio of the concentration of a component in the organic phase to its concentration in the aqueous phase, is a key parameter in liquid-liquid extraction.
The partition coefficient depends on the chemical nature of the solvents and the components being extracted. For example, polar components are generally more soluble in water, while nonpolar components are more soluble in organic solvents. In addition, the partition coefficient is affected by the pH of the aqueous phase, the temperature of the system, and the presence of other solutes.
Techniques of Liquid-Liquid Extraction
There are two main techniques of liquid-liquid extraction ?
Batch extraction.
Continuous extraction.
Batch Extraction
In batch extraction, the mixture to be separated is added to a container containing the two immiscible solvents. The mixture is then agitated or stirred to ensure complete mixing. After a period of time, the two phases are allowed to separate, and the upper and lower layers are removed using a separating funnel or a pipette.
Batch extraction can be performed in a single stage or in multiple stages, depending on the complexity of the mixture and the desired degree of separation. In a single-stage extraction, the mixture is only contacted once with the two-phase system, while in a multiple-stage extraction, the mixture is contacted with the two-phase system several times, each time with fresh solvent.
Continuous Extraction
Continuous extraction involves the continuous flow of the mixture to be separated into a series of extraction columns, where it is contacted with the two immiscible solvents. The columns are typically packed with a solid support material, such as silica gel or ion exchange resin, which enhances the contact between the two phases and improves the efficiency of the extraction.
Continuous extraction is often used when large quantities of material need to be processed or when a high degree of separation is required.
Applications of Liquid-Liquid Extraction
Liquid-liquid extraction is a versatile separation technique that has a wide range of applications in the chemical, pharmaceutical, and environmental industries. Some of the common applications of LLE are discussed below.
Separation
Separation of Impurities
One of the most common applications of LLE is the removal of impurities from a mixture. In many chemical and pharmaceutical processes, impurities can interfere with the quality or effectiveness of the final product. LLE can be used to remove impurities, such as by-products, unreacted starting materials, or catalysts, from a reaction mixture. The impurities can be partitioned into the aqueous phase or the organic phase, depending on their relative solubilities, and removed from the mixture by simple separation.
Concentration of Products
LLE can also be used to concentrate a desired product from a mixture. For example, in the production of natural products or drugs, the target compound may be present in low concentrations in a complex mixture. LLE can be used to selectively extract the target compound into the organic phase, leaving the other components in the aqueous phase. The organic phase can then be evaporated to concentrate the target compound, which can be further purified by other techniques, such as chromatography.
Isolation of Specific Compounds
LLE is often used to isolate specific compounds from complex mixtures, such as natural products, plant extracts, or environmental samples. In these cases, LLE is often combined with other separation techniques, such as chromatography or distillation, to obtain highly pure compounds. LLE can also be used to fractionate mixtures into different components based on their relative solubilities. For example, a mixture of fatty acids can be separated into saturated and unsaturated fractions by LLE, which can then be further purified by other techniques.

Extraction of Metals
LLE is widely used in the extraction of metals from ores, concentrates, and industrial waste streams. In these cases, LLE is often used to selectively extract the metal ions into an organic phase, leaving the other ions in the aqueous phase. The organic phase can then be stripped of the metal ions, which can be further processed to obtain the pure metal or its compounds. LLE is also used in the treatment of industrial wastewater, where it can be used to remove heavy metals or other toxic substances.
Extraction of Organic Compounds from Environmental Samples
LLE is commonly used in environmental analysis to extract organic compounds, such as pesticides, polycyclic aromatic hydrocarbons (PAHs), or pharmaceuticals, from water, soil, or sediment samples. In these cases, LLE is often combined with other sample preparation techniques, such as solid-phase extraction or derivatization, to improve the efficiency and selectivity of the extraction. The extracted compounds can then be analyzed by chromatography or other analytical techniques.
Conclusion
Liquid-liquid extraction is a powerful separation technique that has a wide range of applications in the chemical, pharmaceutical, and environmental industries. The technique is based on the principle of differential solubility of components in two immiscible solvents and can be performed using batch or continuous techniques.
LLE can be used for the removal of impurities, the concentration of a desired product, or the isolation of specific compounds from a complex mixture. The versatility of LLE makes it an essential tool in many industrial processes and analytical methods.


