Article Categories
- All Categories
-
 Data Structure
Data Structure
-
 Networking
Networking
-
 RDBMS
RDBMS
-
 Operating System
Operating System
-
 Java
Java
-
 MS Excel
MS Excel
-
 iOS
iOS
-
 HTML
HTML
-
 CSS
CSS
-
 Android
Android
-
 Python
Python
-
 C Programming
C Programming
-
 C++
C++
-
 C#
C#
-
 MongoDB
MongoDB
-
 MySQL
MySQL
-
 Javascript
Javascript
-
 PHP
PHP
-
 Economics & Finance
Economics & Finance
What is Aluminium Anodizing? – Working Principle, Process
What is Anodizing?
The process of deposition of an oxide film on a metal surface is known as anodizing. The anodizing is an electrolytic passivation process used to increase the thickness of the natural oxide layer on the surface of metal workpieces.
Anodizing of Aluminium
Aluminium alloys are anodized for the following reasons −
To increase the corrosion resistance.
To increase the surface hardness.
To allow coloring or dyeing.
To improve lubrication.
To improve adhesion.
When the pure aluminium is exposed to air at room temperature or any other gas containing oxygen, it self-passivates by forming a surface layer of amorphous aluminium oxide of thickness of 2 to 3 nm. This layer provides very effective protection against corrosion.
Aluminium alloys typically form a thicker oxide layer of thickness of 5 to 15 nm. But, this layer tends to be more susceptible to corrosion. Therefore, the aluminium alloys are anodized to increase the thickness of this oxide layer for corrosion resistance.
Working Principle of Aluminium Anodizing
Anodizing of aluminium works on the principle of electrolysis. In this process, an anodized tank is filled with a suitable electrolyte. In this tank, the aluminium product is suspended to expose most of the surface to the electrolyte.
Now, the plates of suitable element (usually aluminium or lead) are placed in the tank and the circuit between the cathode and anode is complete through a power source.
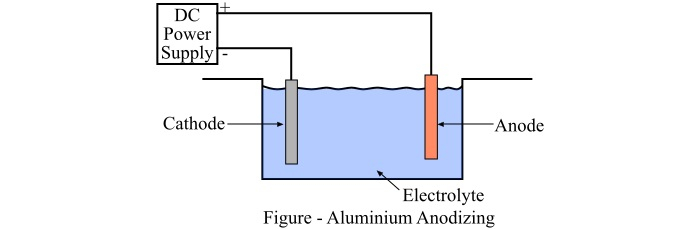
The aluminium product is connected to the positive terminal and the plates are connected to the negative terminal of the power source. As the circuit is completed, the current passes through it.
The strength and duration of flow of electric current will determine the thickness of the aluminium oxide layer on the anodized aluminium product.
Aluminium Anodizing Process
Aluminium anodizing involves the following steps −
Step 1 - Cleaning
In this step, the surface of the aluminium product is cleaned. For cleaning, it is exposed to acidic or alkaline cleaning agents to clean the grease and dirt from the surface.
Step 2 - Pre-treatment
In this step, any surface imperfections are eliminated so that a visible finish with a clean and smooth surface is obtained. The pre-treatment of aluminium is done by using two main processes viz. − brightening and etching.
Step 3 - Anodizing
This step involves the anodizing of the aluminium. For the aluminium anodizing process, the sulfuric acid is used as the electrolyte. There are various alternatives of sulfuric acid which are − organic acid, borate, tartrate, phosphoric acid, etc.
Step 4 - Coloring
In this step, color is added to the anodized aluminium by employing a suitable method. Different colors require different methods, but two most popular methods are electro-coloring and dyeing.
Step 5 - Sealing
It is the final step in the aluminium anodizing process. Sealing prevents the water leakage and improves the corrosion resistance of the anodized aluminium product. There are three methods for the sealing viz. − hot method, cold method and a combination of the two.


